First Law of Thermodynamics
First Law of Thermodynamics: Overview
This topic covers concepts such as Sign Convention in Thermodynamics, First Law of Thermodynamics, First Law of Thermodynamics in Isochoric Process, Heat in Isochoric Process, First Law of Thermodynamics in Isobaric Process, etc.
Important Questions on First Law of Thermodynamics
An ideal gas is allowed to expand freely against vacuum in a rigid insulated container. The gas undergoes
When heat is supplied to the system then is ____ and is ____ when heat is extracted from the system. Choose the appropriate option.
The ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
When a system is taken from state to state along the path , it is found that and . Along the path , and along the path is
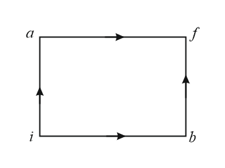
The diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from is and heat is given to the system. The change in internal energy between and is
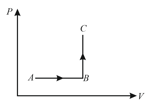
A sample of liquid in a thermally insulated container (a calorimeter) is stirred for , by a mechanical linkage to a motor in the surrounding, for this process:
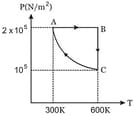
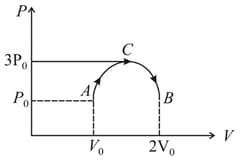
Two moles of a monatomic ideal gas are taken through a cyclic process shown on the diagram. The process is represented as = constant. If the efficiency of the given cyclic process is , then what is the value of ?
A spherical bubble inside water has radius Take the pressure inside the bubble and the water pressure to be The bubble now gets compressed radially in an adiabatic manner so that its radius becomes For the magnitude of the work done in the process is given by , where is a constant and The value of is _______ correct to nearest integer.
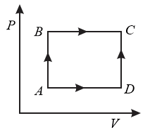
A thermodynamic cyclic process has been depicted in the adjoining diagram. It is given that the heat absorbed and work done by the system along the path is and , respectively. If the amount of work done along the path is , then what will be the amount of heat absorbed in along the same path?
of rigid diatomic gas performs a work of when heat is supplied to it. The molar heat capacity of the gas during this transformation is The value of is ________ .
A monatomic ideal gas undergoes a process . The heat given to the gas is,
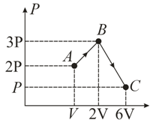
For free expansion of the gas which of the following is true
A -mol ideal gas system undergoes an adiabatic process where it expands and does of work on its environment. How much heat is received by the system?
Which of the following is equivalent to the principle of energy conservation?
An ideal gas undergoes change in its state from the initial state to the final state via two possible paths as shown below. Then,
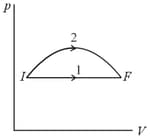
One mole of an ideal monatomic gas is taken through a semicircular thermodynamic process shown in the diagram. The heat supplied to the system is
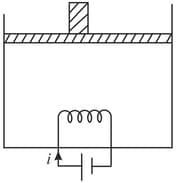
A resistance coil, connected to an external battery, is placed inside an adiabatic cylinder (whose axis is vertical). The cylinder is fitted with a frictionless piston of mass and trapping an ideal mono-atomic gas. A constant current flows through the coil of resistance Neglect the atmospheric pressure. It is observed that the piston moves slowly and with constant speed. That constant speed is
The workdone on the system in changing the state of a gas adiabatically from equilibrium state to equilibrium state is . If the gas is taken from state to through another process in which the net heat absorbed by the system is , then the net workdone by the system in the later case is
An electric heater supplies heat to a system at a rate of If the system performs Work at a rate of joules per second, the rate at which internal energy changes is
Two cylinders and of equal capacity are connected to each other via a stopcock. A contains an ideal gas at standard temperature and pressure. is empty. Then the entire system is thermally insulated. The stopcock is suddenly opened. After the gas reaches equilibrium the temperature of the gas measured. Which of the following is correct?
